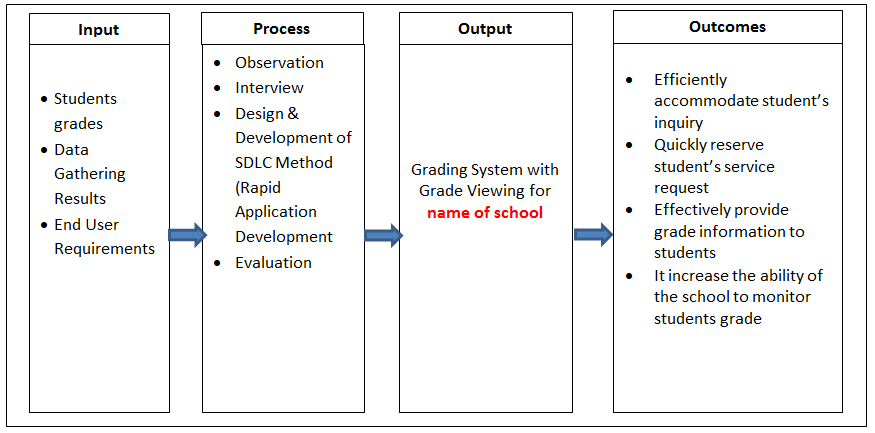
On November 21, the US FDA announced that one lot of Highly Concentrated Potassium Chloride Injection, 10 mEq per 100 mL by Baxter International Inc., is recalled due to mislabeled overpouch. In light of this news, AttorneyOne, a recognized authority on law, updated the website providing detailed information to consumers.
(PRWeb November 25, 2014)
Read the full story at http://www.prweb.com/releases/2014/11/prweb12351651.htm